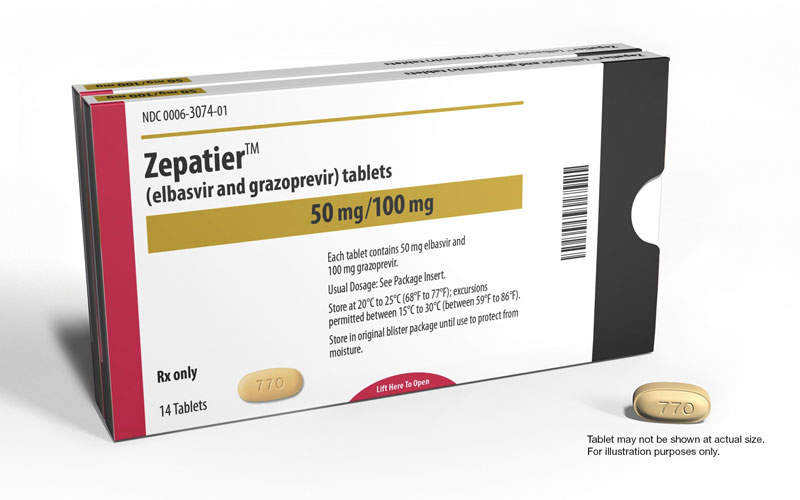
Zepatier is a once-daily oral medicine comprising 50mg of non-structural protein 5A (NS5A) replication complex inhibitor elbasvir and 100mg of NS3/4A protease inhibitor grazoprevir.
It is indicated for the treatment of patients with chronic hepatitis C virus (HCV) genotype 1 (GT1) that have end-stage renal disease, and patients with chronic HCV GT4.
The new drug application (NDA) for Zepatier was submitted by Merck to the US Food and Drug Administration (FDA) on 28 May 2015. The FDA granted breakthrough therapy designation status for the drug in April of the same year, and approved the drug for the treatment of patients with chronic HCV GT1 and GT4 infections on 28 January 2016.
Merck submitted a license application for Zepatier in Europe in July 2015. The drug received positive opinion for approval from the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) in May 2016.
The European Commission (EC) granted marketing authorisation to Zepatier in July 2016. The drug was launched into the European market in November 2016.
Zepatier was also approved in Canada in February 2016.
Hepatitis C virus GT1 and GT4 infection
Hepatitis C infection is a liver disease caused by hepatitis C, a blood borne virus. The infection may range from an acute mild illness to a chronic lifelong illness.
HCV is a small, enveloped, single-stranded, positive-sense ribonucleic acid (RNA) virus that occurs in seven major genotypes, which are classified from one to seven. It causes inflammation of the liver and leads to decreased liver function or failure.
The infection is typically asymptomatic until later stages, which takes several years. The chronic viral infection is characterised by cirrhosis, which is associated with complications such as bleeding, jaundice, fluid accumulation in the abdomen, infections or liver cancer.
Approximately three million Americans are estimated to have HCV. Of these, GT1 is most common and GT4 is least common.
Zepatier’s (elbasvir and grazoprevir) mechanism of action
Zepatier is a combination of a NS5A replication complex inhibitor elbasvir and a NS3/4A protease inhibitor grazoprevir. Elbasvir prevents replication of viral cells by interacting with the NS5A protein on HCV replicon cells, which are responsible for reducing human interferon antiviral activity.
Grazoprevir stops viral replication by binding to the NS3/4A protease, a serine protease domain that is essential for viral poly-protein maturation and neutralisation of the host’s innate antiviral immunity for HCV.
Clinical trials on Zepatier
The FDA approved Zepatier based on results obtained from the C-SCAPE (Phase II), C-SALVAGE (Phase-II), C-SUFFER (Phase IIb / III), C-EDGE (Phase III), C-EDGE TN (Phase III), C-EDGE CO-INXFN (Phase III) and C-EDGE TE (Phase III) clinical trials.
The open-label, randomised, double-blind, placebo-controlled clinical trials enrolled 1,373 patients with chronic HCV GT1 or GT4 infection to demonstrate the safety and efficacy of Zepatier by assessing the rate of sustained virologic response 12 weeks after completion of treatment (SVR12).
The trials conducted on HCV GT1 patients were C-EDGE TN, C-EDGE CO-INFXN, C-SUFFER, C-EDGE TE and C-SALVAGE. The trials conducted on HCV genotype 4 infected patients were C-SCAPE, C-EDGE TN, C-EDGE CO-INFXN and C-EDGE TE.
Merck also conducted three more Phase II trials named C-CREAST 1, C-CREAST 2 and C-WORTHY on HCV GT1, GT2 and GT3 patients, respectively.
The drug’s safety and efficacy with or without ribavirin were evaluated in clinical trials on 1,373 chronic HCV GT1 or GT4 patients with and without cirrhosis.
Subjects were administered Zepatier with or without ribavirin once daily for either 12 or 16 weeks. Overall SVR rates were observed to be from 94% to 97% in GT1 infected subjects, while the range in GT4 infected subjects was from 97% to 100%.
Fatigue, headache and nausea were the most common side effects of Zepatier without ribavirin, while those in Zepatier with ribavirin were anaemia and headaches.