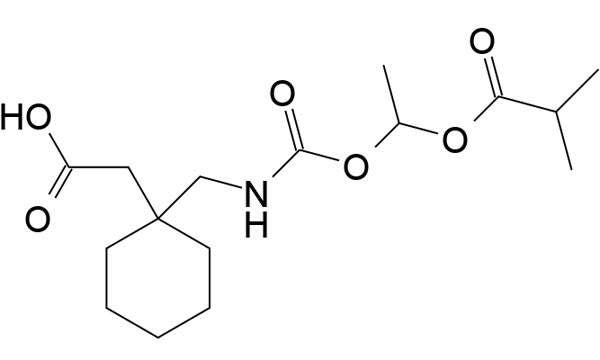
Durbin is a specialist provider of comparator drugs for clinical trials. In addition to comparator drugs, Durbin also provides ancillary supplies and placebos. Durbin brings global sourcing expertise, transparent pricing and global custom distribution to the supply of comparators and ancillaries for clinical trials.
Durbin has over 45 years’ experience in sourcing pharmaceuticals and distributing them worldwide. Durbin combines this heritage with expertise in supplying comparators to pharmaceutical companies, both directly and via third parties, to bring a difference to the market.
Global sourcing expertise in comparator drug supply
Durbin buys direct from manufacturers and wholesalers. We have extensive and close relationships with all the top 30 pharmaceutical companies across Europe and North America. Sourcing from manufacturers and wholesalers guarantees pedigree and a reliable audit trail. We also supply placebos and ancillary items such as infusion pumps, non-PVC administration sets and infusions. We offer:
- Any pack size
- Single batch number
- Any quantity
- Same brands from multiple markets
Transparent pricing for clinical trial supply
Durbin is committed to openness throughout the supply process. We will share with you our cost price and negotiate with you a mutually acceptable profit margin. Sourcing generically can offer up to 80% discount on branded list price. Durbin works closely with all generic companies to negotiate substantial discounts for our clients. We offer:
- Transparency in costing
- Invoices provided for all costs
- Negotiated profit margins
- Partnership approach
Global bespoke distribution for clinical trial comparators
Durbin’s expertise in distribution ensures we can deliver clinical trial comparators to trial centres across the world, including China, Russia and India. We can deliver any amount and supply Europe within 24 hours. Having multi-lingual account managers enables us to negotiate directly with importation authorities to reduce duty payments and delays. The languages spoken by our team include Cantonese, Mandarin, Russian, Hindi, Spanish, French and German.
Benefits of our clinical trial comparator supply service include:
- Frequent deliveries to over 180 countries
- Cold chain and ambient supply chain
- Fully tracked deliveries including temperature control
- Specialised logistics team
Expertise in oncology, cold chain and high-value comparator drugs
Durbin has built a reputation of being able to source and supply oncology, cold chain and high-value comparator drugs quickly, efficiently and at lower than normal costs. We will devise sourcing and distribution strategies to lower the costs of clinical trial supply.
Clinical trial supply partners to the global pharmaceutical industry and specialised biotech companies
Durbin has extensive contracts and partnerships with the top 30 global pharmaceutical companies and research-based biotech companies. We can advise on all areas of clinical trial supply and logistics.